Find a Treatment
25 Treatments
25 Treatments
-
Body Areas
Stomach Breasts Face Nose Butt Body Hair Teeth&Gums Neck Lips Eyes
-
Concerns
Stomach Stomach Stomach
-
Categories
Stomach Stomach Stomach
-
Worth It Rating
Body Test Stomach Stomach
-
Body Areas
Stomach Breasts Face Nose Butt Body Hair Teeth&Gums Neck Lips Eyes
-
Concerns
Stomach Stomach Stomach
-
Categories
Stomach Stomach Stomach
-
Worth It Rating
Body Test Stomach Stomach






















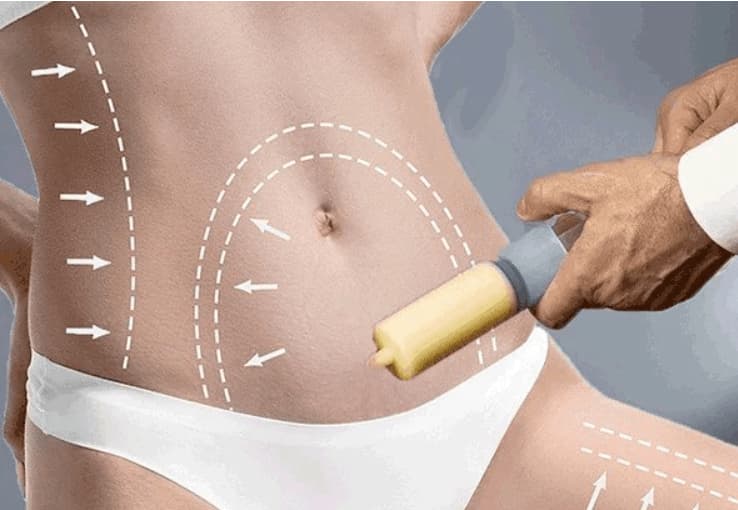






.webp)






.webp)
 19 Reviews
19 Reviews
 Average Cost $ 1500
Average Cost $ 1500